Inorganic Materials Chemistry

Our research areas:
- Atomic/molecular layer deposition
- New (multi)functional materials
- Materials for next-generation energy technologies

Pioneering new-material research for the next-generation sustainable energy and nanoelectronics technologies, including novel functional materials for batteries, thermoelectrics, solar cells, superconductors, spintronics, etc. Current focus on hybrid materials where inorganics are combined down to molecular-level precision with e.g. organic molecules, polymers and biomaterials. As unique new-material synthesis tools, we employ atomic/molecular layer deposition (ALD/MLD) and ultra-high-pressure (HP) techniques.
Examples of Exciting Material Families:
High-Tc Superconductive Copper Oxides:
The purpose of our research on novel multi-layered copper oxide materials is to expand the number of superconductive and related copper-oxide phases. The impact is expected to be two-fold: (i) to gain deeper understanding on the general rules that determine the high-Tc superconductivity characteristics of layered copper oxides, and (ii) to find technologically attractive novel superconductors. Recently we discovered among others three entirely new homologous series of superconductive copper oxides, Ba2Can-1CunO2+2n(n = 2, 3, 4), (HmO2m-2)Ba2Can-1CunO2+2n (n = 2, 3, 4) and (Cu,Mo)Sr2(Ce,R)sCu2O5+2s (R=La~Yb; s = 1, 2, 3, 4, 5, 6). All these series exhibit exciting chemical features not earlier seen for other high-Tc superconductors: peroxide-type oxygen in the first case, charge-reservoir water in the second case and in the third case a ~(3+2s)Å thick fluorite-structured (Ce,R)-[O2-(Ce,R)]s-1 block between adjacent CuO2 planes. Through careful studies on the four members of latter homologous series, (Cu,Mo)Sr2(Ce,Y)sCu2O5+2s (s = 2, 3, 4, 5), a rather amazing conclusion could be made that Tc remains essentially unaffected (at 53 K) upon inserting additional fluorite-structured layers between the two CuO2 planes as long as the hole-doping level of the planes is kept constant
- Grigoraviciute, M. Karppinen, T.S. Chan, R.S. Liu, J.M. Chen, O. Chmaissem & H. Yamauchi, Electronic structures, hole-doping and superconductivity of the s = 1, 2, 3 and 4 members of the (Cu,Mo)-12s2 homologous series of superconductive copper oxides,Journal of the American Chemical Society 132, 838 (2010).
- W. Gao, K. Oishi, H. Suematsu, H. Yamauchi & M. Karppinen, Homologous series of (Cu,Mo)Sr2(Y,Ce)sCu2O5+2s+δ copper-oxide superconductors: Synthesis of members up to s = 6, Solid State Communications 151, 1400 (2011).
- O. Chmaissem, I. Grigoraviciute, H. Yamauchi, M. Karppinen & M. Marezio, Superconductivity and oxygen ordering correlations in the homologous series of (Cu, Mo)Sr2(Ce,Y)sCu2O5+2s+d, Physical Review B 82, 104507 (2010).
The first six members of the homologous series of (Cu,Mo)-12s2 or (Cu,Mo)Sr2(Ce,Y)sCu2O5+2s superconductive copper oxides.
Oxide Thermoelectrics:
Thermoelectric materials provide us with a highly potential means of energy harvesting as through such materials it is possible to produce electricity directly from various waste heat sources. The merit of thermoelectric devices over conventional power generators is that they are completely free from CO2 generation. The major obstacle in putting this attractive phenomenon into large-scale practical uses is the energy-conversion efficiency of the thermoelectric material itself. The misfit-layered cobalt oxide family, [MmA2Om+2±d]q[CoO2], possesses high potential particularly for high-temperature thermoelectric applications most strongly demanded by the industry. Our research aims at discovering the not-yet-known members of this family through manipulating the number and sequence of the different atomic layers in the unit cell and then tailoring the most promising members of the family for optimized performance based on the state-of-the-art concept of nanostructuring. The nanostructures should be designed with dimensions smaller than the phonon mean free path but larger than that for the electron or the hole. In such a situation thermal conductivity would be reduced whereas electrical conductivity should remain intact. Here we will employ the ALD technique for the nanostructuring with the help of both natural and artificial nanotemplates by taking full advantage of the various unique features of the ALD technique, such as atomic level control of the film thickness, perfect step coverage and large-area uniformity.
- J. Lybeck, M. Valkeapää, S. Shibasaki, I. Terasaki,H. Yamauchi & M. Karppinen, Thermoelectric properties of oxygen-tuned ALD-grown [Ca2CoO3]0.62[CoO2] thin films, Chemistry of Materials 22, 5900 (2010).
- H. Yamauchi, L. Karvonen, T. Egashira, Y. Tanaka & M. Karppinen, Ca-for-Sr substitution in the thermoelectric [(Sr,Ca)2(O,OH)2]q[CoO2] misfit-layered cobalt-oxide system, Journal of Solid State Chemistry 184, 64 (2011).
- L. Karvonen, S. Yoon, P. Hug, H. Yamauchi, A. Weidenkaff & M. Karppinen, The n = 3 member of the SrCoO(3n-1)/n series of layered oxygen-defect perovskites, Materials Research Bulletin 46, 1340 (2011).
Nonstoichiometric oxides for SOFCs and oxygen storage:
Oxygen-nonstoichiometric oxide materials are useful in a variety of applications. Firstly, presence of vacant oxygen sites makes it possible for oxide ions to move with a reasonable speed in the crystal lattice; this is utilized in applications ranging from the solid-oxide fuel cells to gas-separation membranes and oxygen sensors. Another important aspect is that nonstoichiometric oxides often allow reversible oxygen charging and discharging, i.e. oxygen storage. Recently we revealed excellent oxygen absorption/ desorption characteristics for the YBaCo4O7+d compound. Now we are working on to tailor this material such that it may be put into practical use; our YBaCo4O7+d-type materials are for instance believed to be highly relevant candidates to be utilized for perfect separation of H2 gas from O2 gas when yielded through decomposition of water by means of photocatalysts or as a new type of electrolyte in SOFCs. We also search for novel anode materials for SOFCs. The present material of choice, i.e. NiO/electrolyte composite, performs well for pure H2, but not for natural gas as a fuel. This is because carbon deposition occurs on the NiO anode surface due to CO2 contained in natural gas. Our goal is to develop new anode materials that are intact in natural gas (without any efforts to purify it). One of the material family investigated is the double-perovskite oxides derived from the Sr2MgMoO6 phase [J.B. Goodenough et al., Science 312, 254 (2006)].
- S. Vasala, M. Lehtimäki, Y.H. Huang, H. Yamauchi, J.B. Goodenough & M. Karppinen, Degree of order and redox balance in B-site ordered double-perovskite oxides,Sr2MMoO6-δ (M = Mg, Mn, Fe, Co, Ni, Zn), Journal of Solid State Chemistry 183, 1007 (2010).
- S. Vasala, M. Lehtimäki, S.C. Haw, J.M. Chen, R.S. Liu, H. Yamauchi & M. Karppinen, Isovalent and aliovalent substitution effects on redox chemistry of Sr2MgMoO6-d SOFC-anode material, Solid State Ionics 181, 754 (2010).
- S. Vasala, H. Yamauchi & M. Karppinen, Role of SrMoO4 in Sr2MgMoO6 synthesis, Journal of Solid State Chemistry 184, 1312 (2011).
- M. Lehtimäki, H. Yamauchi & M. Karppinen, Oxygen absorption/desorption characteristics of Pb2CuSr2RCu2O8+δ, Solid State Ionics 182, 71 (2011).
- S. Räsänen, O. Parkkima, E.-L. Rautama, H. Yamauchi & M. Karppinen, Ga-for-Co substitution in YBaCo4O7+d: Effect on high-temperature stability and oxygen-storage capacity, Solid State Ionics 208, 31 (2012).
Oxide halfmetals and multiferroics:
Halfmetallic and multiferroic materials are expected to provide us with novel phenomena of high scientific and technological impacts. In a ferromagnetic halfmetal only one of the two spin bands is partially occupied. As a consequence, electrical conduction is due to carriers of one spin direction only, i.e. 100 % spin-polarized carriers. This allows halfmetals to be used in tunneling-type magneto-resistance devices in which the spin-dependent electron transport is sensitively controlled by an external magnetic field. Multiferroics, on the other hand, are materials with mutually coupled magnetic and (di)electric properties. In such materials the magnitude of dielectric constant may be controlled by the magnetic field through a so-called magnetodielectric or magnetocapacitance effect. To date, only a handful of halfmetallic and multiferroic materials have been discovered. Hence, the challenge is to expand the yet limited number of compounds showing these exciting phenomena. Ordered perovskites are highly promising material candidates, such as the A-site ordered magnetodielectric double-perovskite oxide (BiMn3)Mn4O12 recently realized by us through ultra-high-pressure synthesis and the halfmetallic B-site ordered double-perovskite oxide Sr2FeMoO6.
- H. Okamoto, N. Imamura, M. Karppinen, H. Yamauchi & H. Fjellvåg, Crystal structure of the monoclinic and cubic polymorphs of BiMn7O12, Journal of Solid State Chemistry 183, 186 (2010).
- N. Imamura, K. Singh, D. Pelloquin, Ch. Simon, T. Sasagawa, M. Karppinen, H. Yamauchi & A. Maignan, Magnetodielectric response of square-coordinated MnO2 unit in cubic BiMn7O12, Applied Physics Letters 98, 072903 (2011).
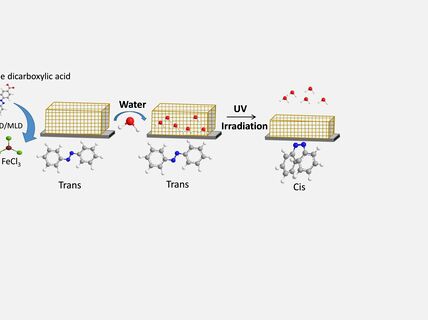
Layer-by-layer deposited inorganic-organic hybrid materials:
There is an obvious need for totally new types of on-demand-designed materials with new/extraordinary combinations of properties. Inorganic-organic hybrid materials are prepared by putting together both inorganic and organic building blocks on a molecular level and are therefore able – in principle – to possess a tailored combination of features traditionally seen for inorganics or organics only. We can imagine e.g. ceramics that are less brittle and easier to process than conventional ceramics, or polymers that are not only flexible but possess diverse electronic, magnetic, dielectric or catalytic properties. In reality, however, a majority of these attractive material characteristics are not necessarily achieved with conventional hybrid materials as their multi-phasic nature may cause severe problems arising from inhomogeneity and weak interactions between the different phases. In the most exciting but little investigated category of hybrid materials, the inorganic and organic entities are not just carefully mixed but are linked together via strong chemical bonds to form coherent single-phase materials. In such nanocomposite materials with strong interactions between the inorganic and organic building blocks multiple unexpected and even seemingly contradictory properties might be simultaneously found. Our goal is to combine the atomic layer deposition (ALD) and molecular layer deposition (MLD) thin-film deposition techniques to produce novel inorganic-organic hybrid materials in a layer-by-layer manner through sequential self-limiting gas-surface reactions with high precision for the film thickness, composition and density as well as polymer-chain dispersity and orientation. As the combination of the two techniques, ALD and MLD, enables the control of abundance of the inorganic and organic constituents in the structure at atomic/molecular level, it is possible to deposit various inorganic-organic alloys and nanolaminates with precisely tuned material properties.
- A. Sood, P. Sundberg, J. Malm & M. Karppinen, Layer-by-layer deposition of Ti-4,4’-oxydianiline hybrid thin films, Applied Surface Science 257, 6435 (2011).
- P. Sundberg, A. Sood, X. Liu, L.-S. Johansson & M. Karppinen, Atomic/molecular layer deposited thin-film alloys of Ti-4,4’-oxydianiline hybrid–TiO2 with tunable properties, submitted (2011).
Thin-film alloys containing precisely controlled proportions of Ti-4,4’-oxydianiline hybrid and TiO2 can be prepared by the combination of ALD and MLD techniques. For such alloys excellent tunability of the film properties such as degree of crystallinity, roughness, refractive index, and hardness is achieved.

Reseach group members: