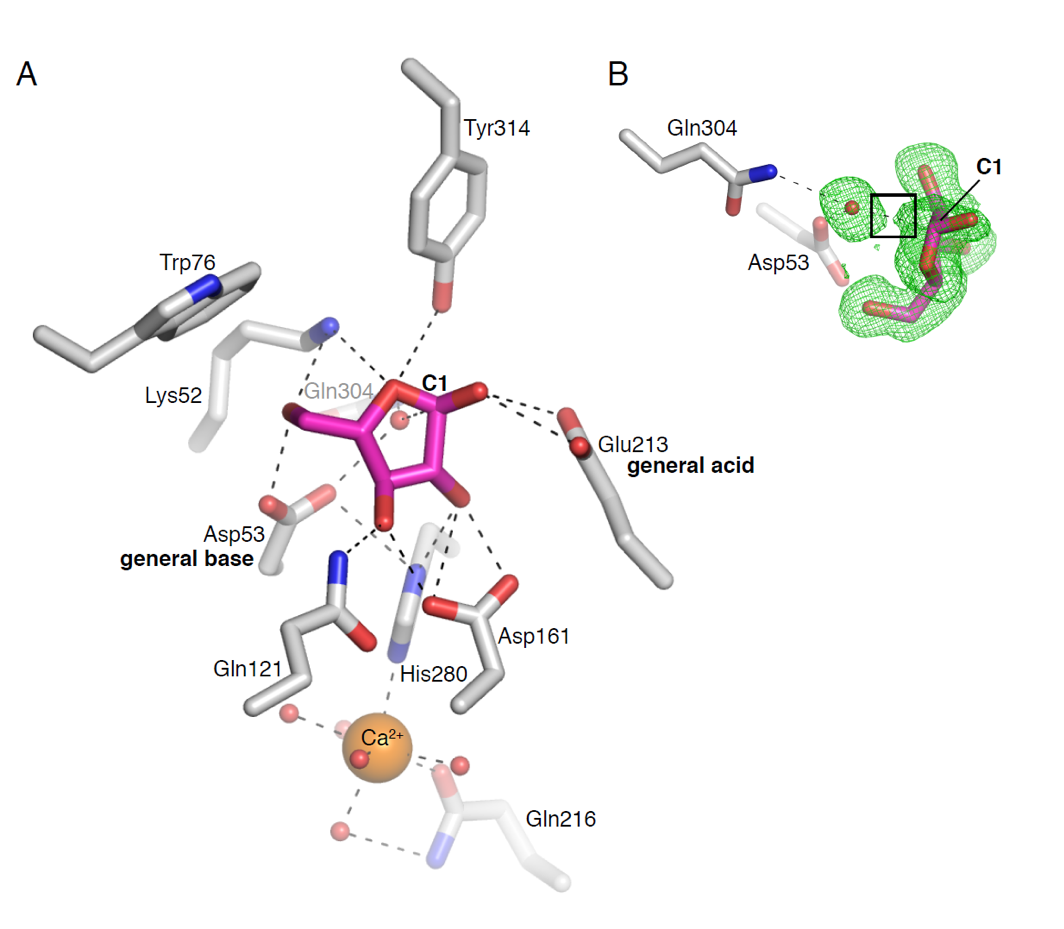
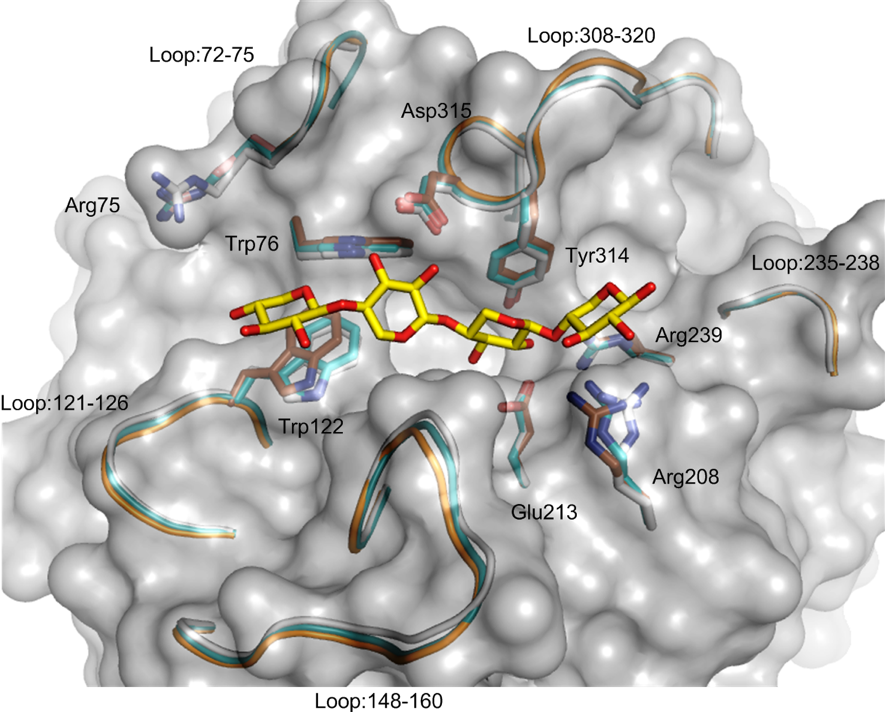
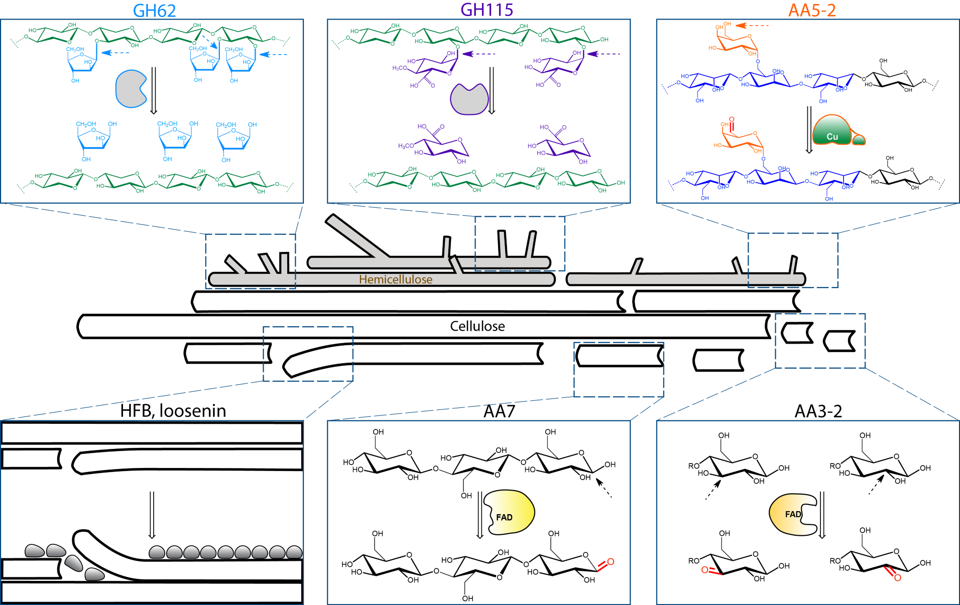
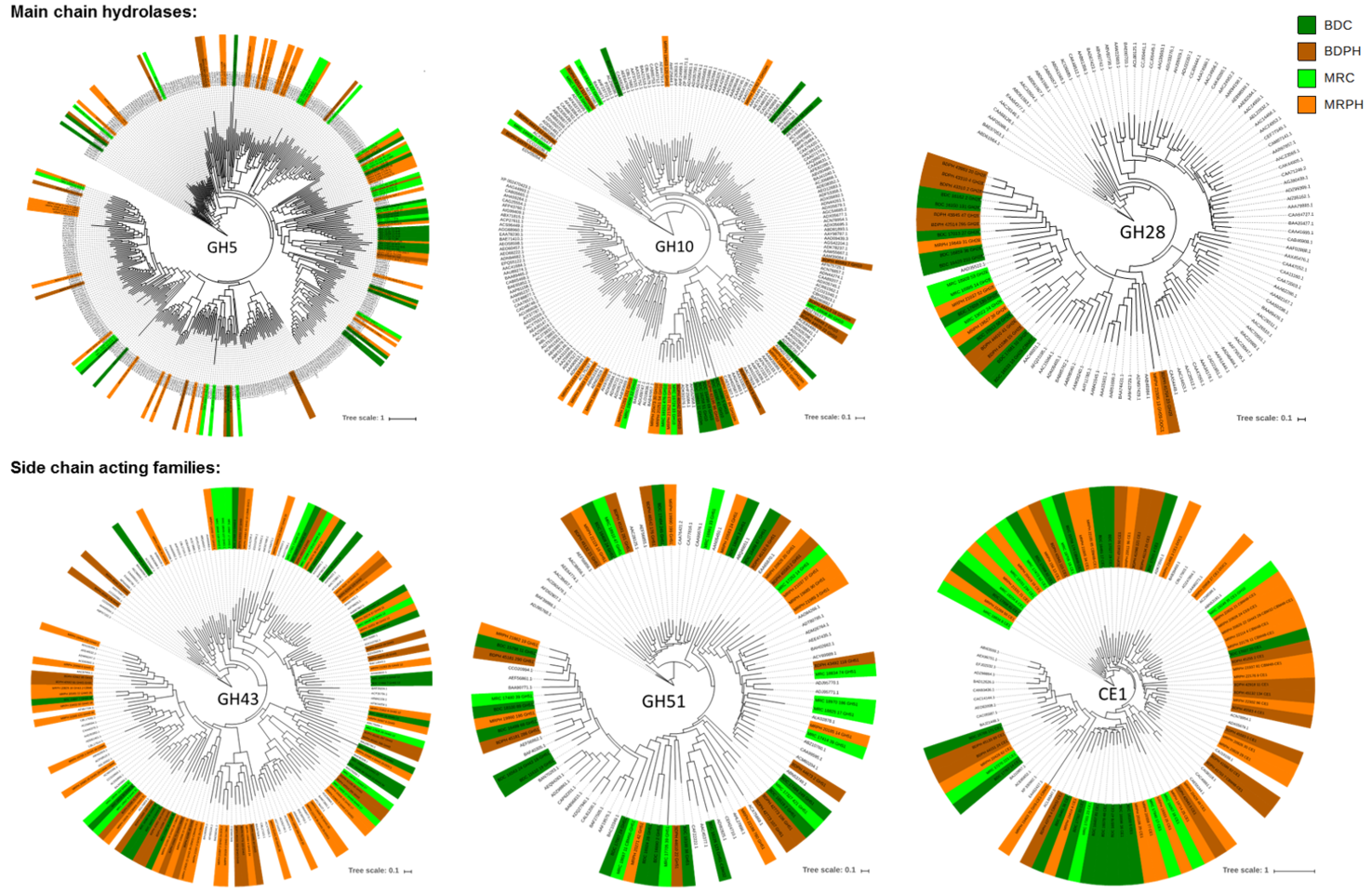
Protein Technology
Our approach applies functional genomics and biophysical methods to develop unique carbohydrate-active enzymes and non-catalytic proteins that control the chemical functionality and assembly of major lignocellulose components.
Research areas:
- Biocatalysts to upgrade renewable bioresources
- Protein Engineering
- Carbohydrate-active enzymes
By using biotechnologies to upgrade biopolymers for multipurpose and sustainable materials, we will:
- leverage environmental and economic benefits that are achieved when retaining naturally fixed CO2 in new bio-based products;
- establish biocatalysts as instruments for bio-based materials engineering;
- realize intact, structural biopolymers as the superior progenitor of high-performance materials.
Examples of ongoing research projects:
- Carbohydrate-active oxidoreductases for bio-based cross linkers
- Biocatalytic cascades for hemicellulose reassembly
- Microbial expansin related proteins for bio-fibre engineering
Research aims to find tools for sustainable utilization of renewable plant resources
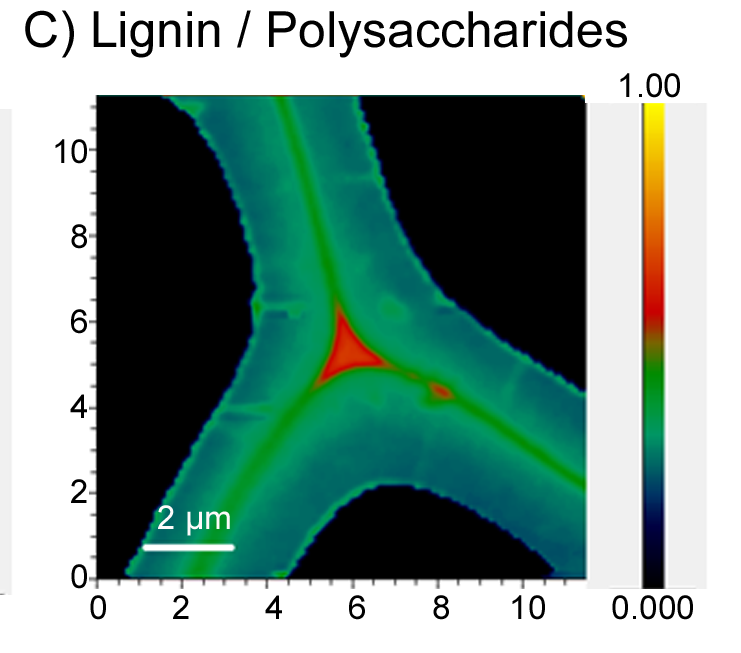
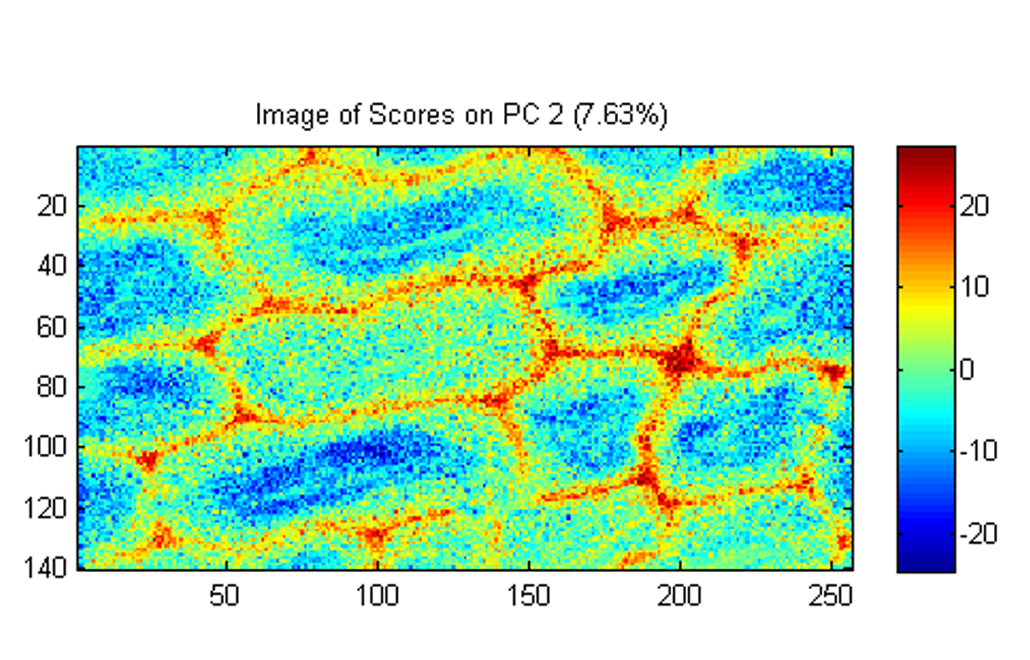
Forest biomass (lignocellulose) is largely made up of three polymers, cellulose, hemicelluloses, and lignin. Today, cellulose fibers are the main product of pulp mills across Finland and globally, while over 50% of the wood biomass resource remains underutilized. Our research focuses on the discovery and development of new proteins that can be used to sustainably synthesize novel, high-value biomaterials from underutilized and renewable plant resources.
Enzymes are tools with advantages
Enzyme catalyzed reactions benefit from four key advantages.
- Specificity that allows predictable modification of complex substrates.
- Exquisite tunability through protein discovery and engineering.
- Operation in mild reaction conditions that can reduce energy costs and undesired transformation of starting materials.
- Biodegradability, which helps to ensure the sustainability of the synthesis process as well as end product.
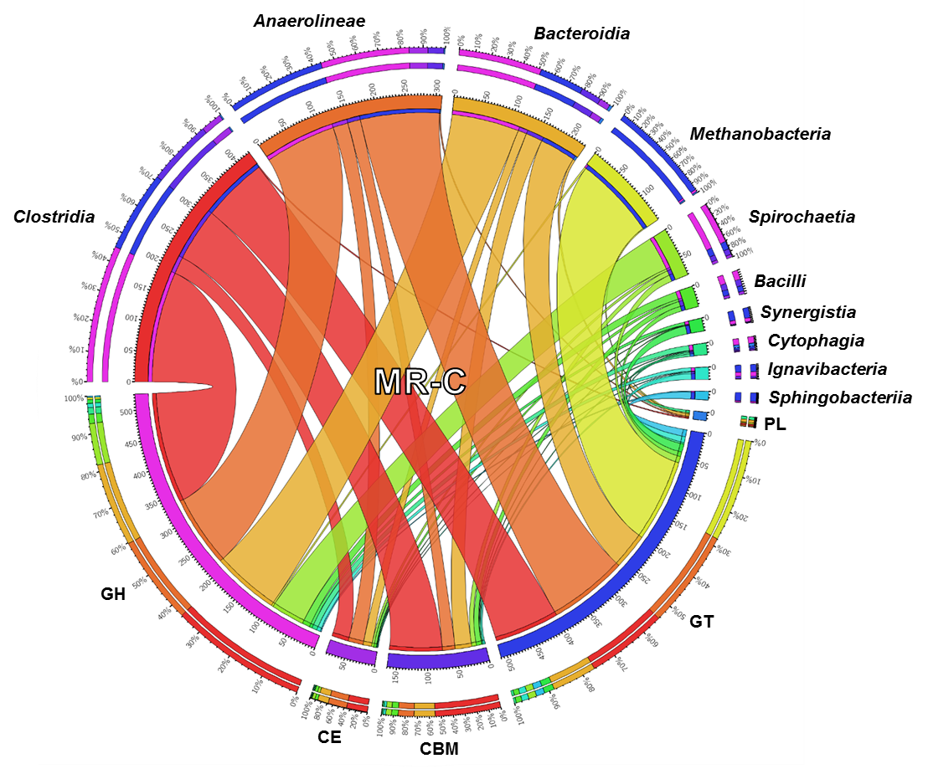
The Protein Technology group mines unexplored genomic data from plant biomass degrading microorganisms to discover new enzymes and non-catalytic proteins able to create valuable, bio-based products from plant polysaccharides. To support this objective, our group also designs and develops new enzyme screens to address the increasing limitations of existing assays.
Research group members: