ABC Seminar: Human Neocortical Neurosolver
When
Where
Event language(s)
In this event, we are committed to Aalto University’s principles for a safer space.
Everyone is welcome to the ABC Seminar, where the newest and coolest of neuroscience is presented and discussed. Entrance is free and you don't need to register.
Our speaker will be Stephanie Jones, the PI of Jones Lab at the Brown university, and she will present their new software tool that helps neuroscientists to bridge the gap between macro (the whole brain or brain regions) and micro scales (neurons and neuronal circuits).
The talk will be followed by coffee and pulla.
Speaker: Prof. Stephanie Jones, Brown University
Title: Human Neocortical Neurosolver: A Neural Modeling Tool to Link Mechanism to Meaning of EEG/MEG Signals
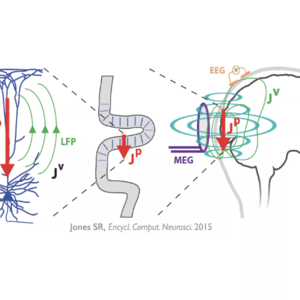
Abstract: Electro- and magneto-encephalography (EEG/MEG) are the leading methods to non-invasively record human neural dynamics with millisecond temporal resolution. However, it can be extremely difficult to infer the underlying cellular and circuit level origins of these macro-scale signals without simultaneous invasive recordings. This limits the translation of MEG/EEG into novel principles of information processing, or into new treatment modalities for neural pathologies. To address this need, we developed the Human Neocortical Neurosolver (HNN: hnn.brown/edu ), a new user-friendly neural modeling tool designed to help researchers and clinicians interpret human imaging data. A unique feature of HNN’s model is that it accounts for the biophysics generating the primary electric currents underlying such data, so simulation results are directly comparable to source localized data. In this talk, I will give an overview of this new tool and describe an application to study the origin and meaning of 15-29Hz beta frequency oscillations, known to be important for sensory and motor function. Our data showed that in primary somatosensory cortex these oscillations emerge as transient high power ‘events’. Functionally relevant differences in averaged power reflected a difference in the number of high-power beta events per trial (“rate”), as opposed to changes in event amplitude or duration. These findings were consistent across detection and attention tasks in human MEG, and in local field potentials from mice performing a detection task. Modeling led to a new theory on the circuit origin of such beta events and suggested beta causally impacts perception through layer specific recruitment of cortical inhibition, with support from invasive recordings in animal models. We are currently expanding applications of the tool to include interpretation of the neural impacts of non-invasive brain stimulation. In total, HNN provides an unpresented biophysically principled tool to link mechanism to meaning of human EEG/MEG signals.






