Bioprocess Engineering

Research themes within the group
- Synthetic Biology and Biological Engineered Living Materials.
- Genome Biology of biomaterial producing bacteria.
- Bioprocess engineering for sustainble and resource-efficient processes.
Ongoing research projects
Engineering surface display in Komagataeibacter to design new ‘smart’ biomaterials (2025 - 2028, CIMANET doctoral school funded by the Research Council of Finland)
This project develops genetic surface-display tools in Komagataeibacter to enable in situ functionalization of bacterial cellulose. By programming cell-surface interactions during material biosynthesis, the work aims to create smart, responsive, and multifunctional biomaterials with applications in engineered living materials.
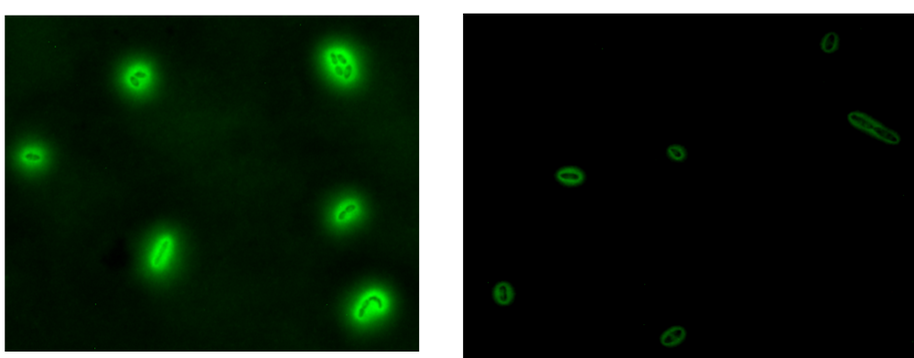
Engineering protein secretion routes in Komagataeibacter rhaeticus to design growing functional materials, ELM 4 PET (2022-2027, Research Council of Finland)
The ELM 4 PET project aims to understand the native protein secretion systems in Komagataeibacter spp. and develop forthright synthetic biology stratagem to integrate heterologous modules for recombinant protein secretion. T
Publications/preprints
Vannas, J., Hannikainen, B., Alexandrino de assis, M., Ledesma-Amaro, R., Ellis, T., Mangayil, R. (2025). Engineering signal peptide-mediated recombinant protein secretion in Komagataeibacter rhaeticus for biological Engineered Living Material applications. bioRxiv. https://doi.org/10.64898/2025.12.15.694344.

Developing in silico pipelines to track IS mobilization in Komagataeibacter via population-level sequencing (2024 - 2027, CYSS doctoral school funded by Jenny and Antti Wihuri Foundation)
The project develops in silico analysis pipelines for population-level sequencing to map and understand genome flexibility of Komagataeibacter spp. under agitated conditions. The resulting frameworks enable predictive assessment of genome stability and engineerability in biomaterial-producing bacteria.
Publications/preprints
Bersanetti, D., Mangayil, R. (2025). Exploring Genome-Wide Mutation Dynamics and Bacterial Cellulose Impairment in Komagataeibacter intermedius Cultivated Under Agitation Stress.bioRxiv. https://doi.org/10.1101/2025.10.09.681338

Metabolic landscape of acetogenic Clostridia under mixotrophic fermentation (2024 - 2027)
This project maps the metabolic landscape of acetogenic Clostridia during mixotrophic fermentation, integrating gas and biomass-derived substrates to understand pathway regulation, guiding process optimization for sustainable biochemical production.
Publications/preprints
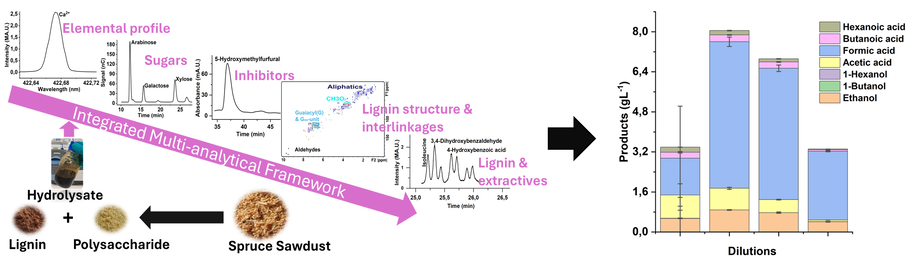
Kumar, S., Nousiainen, P., Kamravamanesh, D., Mangayil, R. (2026). Integrated multi-analytical framework for comprehensive characterization of lignocellulosic hydrolysates for biorefinary applications. Biomass and Bioenergy, 207, 108754. (https://doi.org/10.1016/j.biombioe.2025.108754)
New bacterial processes in the global carbon cycle and in the mitigation of methane emissions (2022-2027)
This project aims in revealing the role of gammaproteobacterial methanotrophs in driving novel microaerobic and anaerobic methane oxidation processes that regulate the consumption of greenhouse gas methane in lakes. The team expects to isolate and characterize new species of methanotrophs and also to develop processes to extend their biotechnological potential to generate value-added products from C1 compounds. The project, funded by the Research Council of Finland, is coordinated by Dr. Antti J Rissanen (Tampere University, Finland) and collaborates with the Rahul Mangayil's BioELM group (Aalto University), The Arctic University of Norway (Norway), Helmholtz-Centre for Environmental Research-UFZ (Germany), University of Basel (Switzerland), Technische Universität Bergakademie Freiberg (Germany), Lammi Biological Station (University of Helsinki), University of Jyväskylä, and University of Eastern Finland.
Our members
Interested in working with us?
Highly motivated MSc or doctoral students or postdoctoral researchers interested in joining the group can contact Dr. Rahul Mangayil (firstname.lastname@aalto.fi).






