Medical Ultrasonics Laboratory (MEDUSA)
Ultrasound provides unconventional approaches to manipulate material from micro-scale, e.g. drug carriers or cells, to macro-scale, e.g. tissue or organs. Ultrasound can be used create force fields through the skin, in a non-destructive manner, to influence targets inside the body from a distance. This allows one to develop non-invasive applications and technologies for advanced medical diagnostics and interventions. Ultrasound can also add value to small minimally invasive medical instruments by activating them based on non-linear ultrasound approaches. Our research has recently contributed to establishing several medical technology companies.
Our current interests include e.g. ultrasound-enhanced fine-needle biopsy (USeFNB), ultrasound-enhanced medical needles, ultrasonic scalpels, ultrasonic drug delivery, and ultrasonic gene transfection. We also conduct basic research on acoustic phenomena that have the potential to be translated to medical applications in the future.

We have multi-national multi-disciplinary team with expertise in physics, biomedical engineering, electrical engineering, acoustics, medicine and business.
The laboratory is headed by Prof. Heikki Nieminen.
News
- 12.12.2025: We would like to congratulate MD Minna Rehell for successfully defending her doctoral thesis entitled ”Open Biopsy and Ultrasound-Enhanced Fine-Needle Aspiration Biopsy in the Head and Neck Region” in our collaborative project between Helsinki University Hospital, University of Helsinki and Aalto University. The Opponent was Professor h.c. Ilpo Kinnunen from the University of Turku. Please, find the thesis here: https://helda.helsinki.fi/items/bd2e8788-1120-4c09-a960-7de70606ad8a
- 21.11.2025: We would like to congratulate Alex Drago González for successfully defending his doctoral thesis entitled ” Dynamic interactions between ultrasound and water-repellent surfaces”. The Opponent was Prof. Michael Baudoin from the University of Lille. Please, find the thesis here: https://aaltodoc.aalto.fi/items/7a477ab0-b61e-462d-b9d2-490c26d4019b
- 06.05.2025: The study by Jussi Kiviluoto et al. entitled "Cavitation activity induced by spring-loaded core needle biopsy devices" has been published today in Scientific Reports. Please, find the article here: https://www.nature.com/articles/s41598-025-97497-z
- 26.03.2025: Our paper by Minna Rehell et al. entitled "Ultrasound-enhanced fine-needle biopsy improves tissue yield in head and neck tumors ex vivo" has been published today in Scientific Reports. Please, find the article here: https://www.nature.com/articles/s41598-025-92610-8
- 12.02.2025: We are happy to announce that the study by Maxime Fauconnier et al. entitled "Fast capillary waves on an underwater superhydrophobic surface" has been published today in Nature Communications. The work was done in collaboration with Prof. Robin Ras laboratory. Please, find the article here:https://doi.org/10.1038/s41467-025-55907-w
- 17.01.2025: We would like to congratulate Yohann Le Bourlout for successfully defending his doctoral thesis entitled ”Ultrasound-enhanced fine-needle aspiration for biopsy: From device development to in vivo human validation”. The Opponent was Prof. Margaret Lucas from the University of Glasgow. Please, find the thesis here: https://aaltodoc.aalto.fi/items/5d30b516-2223-461f-9f97-67b0188bf0d0)
- 01.01.2025: We are pleased to welcome our new Doctoral student Jussi Kiviluoto to join MEDUSA.
- 01.10.2024: We are pleased to welcome our new Doctoral student Cain Santhan to join MEDUSA.
- 06.08.2024: We would like to congratulate our Doctoral Researcher Alex Drago-Gonzaléz for receiving the NBE Excellent Student Paper Award for the article entitled “Ultrasonic Healing of Plastrons" published in Advanced Science. Please, find the article here: https://doi.org/10.1002/advs.202403028
- 01.07.2024: We are happy to announced that the study by Drago-González et al. entitled "Ultrasonic Healing of Plastrons" has been published today in Advanced Science. Please, find the article here: https://doi.org/10.1002/advs.202403028
- 04.06.2024: The study by Le Bourlout et al. entitled "Ultrasound-Enhanced Fine-Needle Biopsy Improves Yield in Human Epithelial and Lymphoid Tissue" has been published today in Ultrasound in Medicine and Biology. Please, find the article here: https://doi.org/10.1016/j.ultrasmedbio.2024.04.015
- 20.05.2024: We are pleased to welcome our new Post-Doctoral Researcher Daniel Veira-Canle to join MEDUSA.
- 15.03.2024: We would like to welcome our new Post-Doctoral Researcher, William Cailly to join MEDUSA.
- 21.12.2023: We would like to congratulate our Doctoral Researcher Yohann Le Bourlout for receiving the NBE Excellent Student Paper Award for the article entitled “Multi-modal transducer-waveguide construct coupled to a medical needle" published in The Journal of the Acoustical Society of America. Please, find the article here: https://doi.org/10.1121/10.0022326
- 14.12.2023: We are happy to announce that the ultrasound-enhanced fine-needle biopsy (USeFNB)received the Aalto School of Science Innovation of the Year award. Read more here: https://www.aalto.fi/en/news/sci-awards-2023-celebrate-the-people-of-our-school
- 22.11.2023: Our paper by Le Bourlout et al. entitled "Multi-modal transducer-waveguide construct coupled to a medical needle" has been published today in The Journal of the Acoustical Society of America. Please, find the article here: https://doi.org/10.1121/10.0022326
- 6.10.2023: We successfully conducted the first human experiment sampling a tumor using ultrasound-enhanced fine-needle biopsy developed at our laboratory. The new method is expected to improve cancer management in the future. For more information, read the team interview here: https://www.aalto.fi/en/news/hus-performs-the-first-biopsy-in-human-with-an-ultrasonic-medical-needle-developed-at-aalto
- 14.9.2023: We would like to welcome our new Doctoral Researcher Nele van Giel to join MEDUSA.
- 12.10.2022: Our paper by Bunni et al. entitled "Needle bevel geometry influences the flexural deflection magnitude in ultrasound-enhanced fine-needle biopsy" has been published today in Scientific Reports. Please, find the article here: https://doi.org/10.1038/s41598-022-20161-3
- 07.10.2022: Congratulation to D.Sc Emanuele Perra for completing his doctoral study.
- 1.9.2022: We would like to welcome Dr. Thomas Andreae, Post-doctoral Researcher Sanjeev Ranjan and Doctoral Researcher Ona Westerlund to join MEDUSA.
- 6.6.2022: We would like to welcome our new Doctoral Researcher Alex Drago-Gonzalez to join MEDUSA.
- 30.5.2022: Jane and Aatos Erkko foundation has provided us a major grant to work for development of a novel gene delivery method.
- 13.6.2022: Our paper by Perra et al. entitled "An Ultrasonically Actuated Needle Promotes the Transport of Nanoparticles and Fluids" has been published today in The Journal of Acoustical Society of America. Please, find the article here: https://doi.org/10.1121/10.0012190
- 02.6.2022: We are happy to announce that our paper by Perra et al. entitled "An ultrasonically actuated fine-needle creates cavitation in bovine liver" has been published today in The Journal of Acoustical Society of America. Please, find the article here: https://asa.scitation.org/doi/full/10.1121/10.0010534
Recent research
Ultrasound-enhanced fine-needle aspiration biopsy (USeFNAB)
Fine-needle biopsy is a common medical procedure for obtaining a tissue sample for diagnostic purpose. This procedure, broadly used in cancer detection, suffers from obtaining and inadequate sample up to 30% of cases.
We are developing a new method, USeFNB, in which ultrasound is used to actuate the very tip of the needle (Perra et al. Scientific Reports. 2021;11:8234; Le Bourlout et al. IEEE IUS 2020). The non-linear interactions of the needle tip and tissue lead to increase in the amount of tissue by 3-5x compared to fine-needle aspiration biopsy (FNAB) using the same needle. The improvement in tissue quantity is demonstrated in the Figure 1. Importantly, the obtained tissue mass increases, while no major effect on sample quality, as demonstrated by histological assessment. The feasibility of the USeFNAB method has been demonstrated recently (Perra et al. 2021) as summarized in the following:
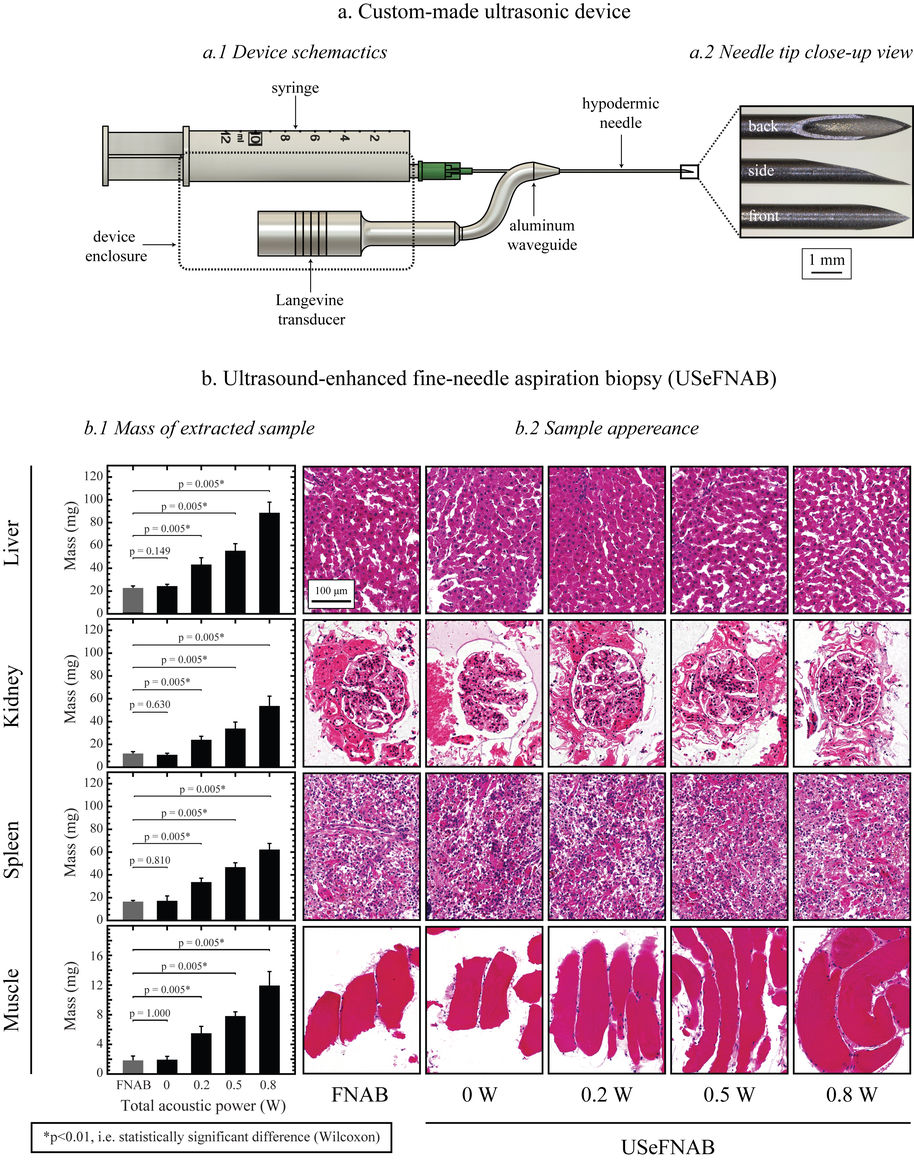
The comparison of the FNAB and USeFNAB procedures with typical tissue mass obtained are exemplified in the following video:
We have further developed the power efficiency of USeFNAB concept to potentially allow batterization of USeFNAB in the future (Le Bourlout et al. 2020). Following optimization, we have reached an electrical-to-acoustic power -efficiency of up to 69%. This result suggests that the developed waveguide structure allows to drive the system with low power electronics permitting miniaturization and portability of the USeFNAB device.
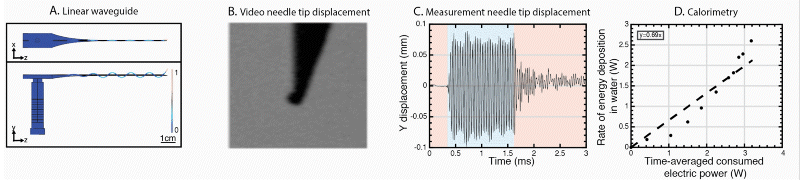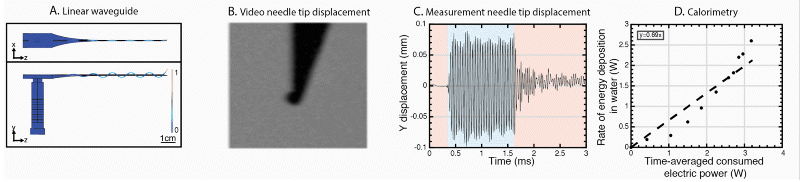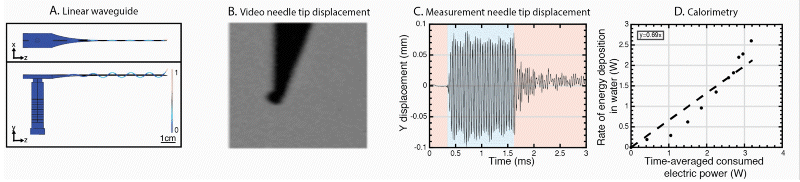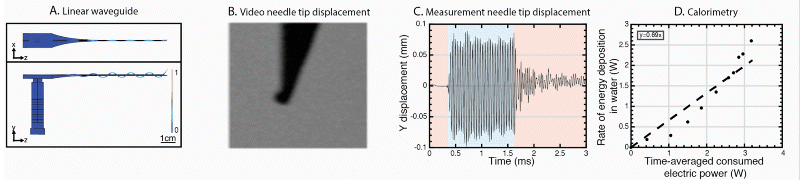
Ultrasound-enhanced medical needle (USeMN)
USeMN could have several potential applications in the future, beyond USeFNAB. Inside liquid, an ultrasonically actuated medical needle can be made to vibrate rapidly producing cavitation bubbles. These bubbles grow and can collapse rapidly. We have recorded accelerations of bubble-water boundary of up to 20 000 G (Perra et al. 2021). With further advancements of the cavitation events localized near the tip of the medical needle could be used for fractionate tissue (histotripsy) such as tumors or even enhance drug delivery by improving tissue permeability. Importantly, the cavitation can be made to exhibit only at the tip of the needle providing predictability to the spatial location of cavitation and, therefore, allows the control safety aspects associated with cavitation. The following high-speed video exemplifies cavitation events at 33 kHz observed near the tip of a USeMN:
While the USeMD is embedded in air, liquid delivered to a tip of a USeMN can be atomized to small droplets using capillary waves delivered to the needle tip (Perra et al. 2021). This approach could be used to deliver and disperse drugs on tissue surfaces within gas-filled bodily cavities, e.g. in the respiratory system. Importantly, we demonstrated that the droplet size can be controlled by the selection of the ultrasound frequency (Perra et al. 2021). Atomization of water at 33 kHz is exemplified in the following high-speed video:
We have also demonstrated that the USeMN is capable of transporting microparticles (Perra et al. 2021). This could be advantageous in various drug and gene delivery applications, since the approach could contribute to the delivery beyond the reach of a traditional needle. The transport of microparticles by 33 kHz ultrasound inside water is demonstrated in the following:
Ultrasonically actuated scalpels
Ultrasonic scalpels are known for their capability of cutting tissue. Usually such technologies employ actuation along the longitudinal axis of the blade. However, transversely oscillating motion has received less attention. In this study, our objective is to broaden understanding of the behaviour of different wave modes emitted from a transversely oscillating scalpel blade, modelled as a wedge embedded in viscoelastic material representing soft tissue. Our simulations demonstrate the different wave modes emitted from the needle. The following 2D velocity surface map shows the vibrating wedge oscillating at 30 kHz on the surface of viscoelastic medium representing soft tissue, captured at a time step 0.66 ms. The x and y components of velocity (m/s) are shown respectively on the left and right:
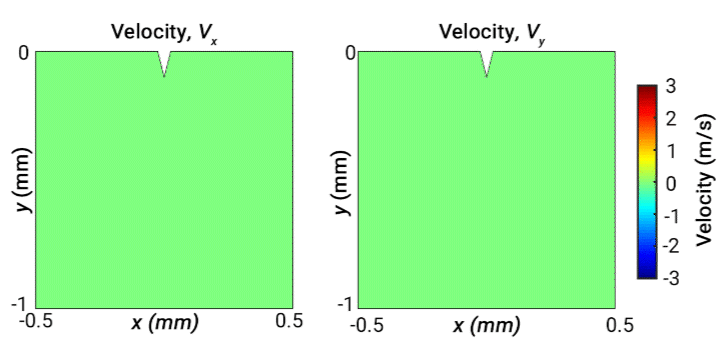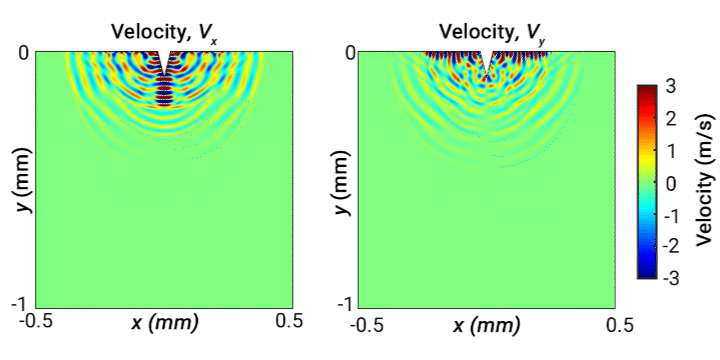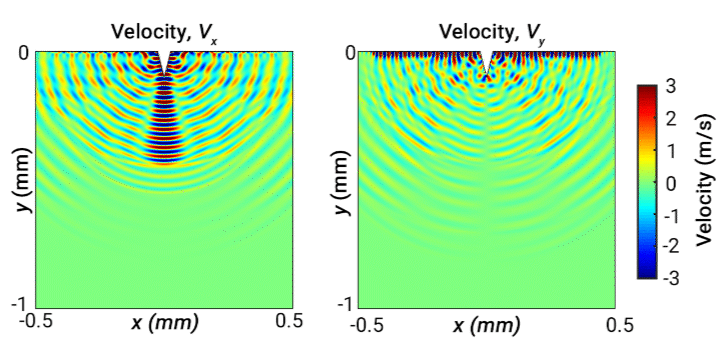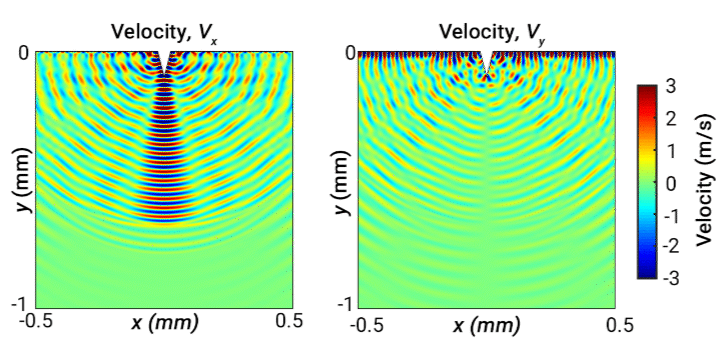
Ultrasonic levitation of particles
Ultrasound can be used to levitate and trap small particles. This approach allows one to manipulate and sort small objects, such as micro-spheres and cells, without touching them. The following example demonstrates how micro-spheres are trapped inside liquid along the axis of the ultrasound propagation as well as along the axis perpendicular to the ultrasound propagation:
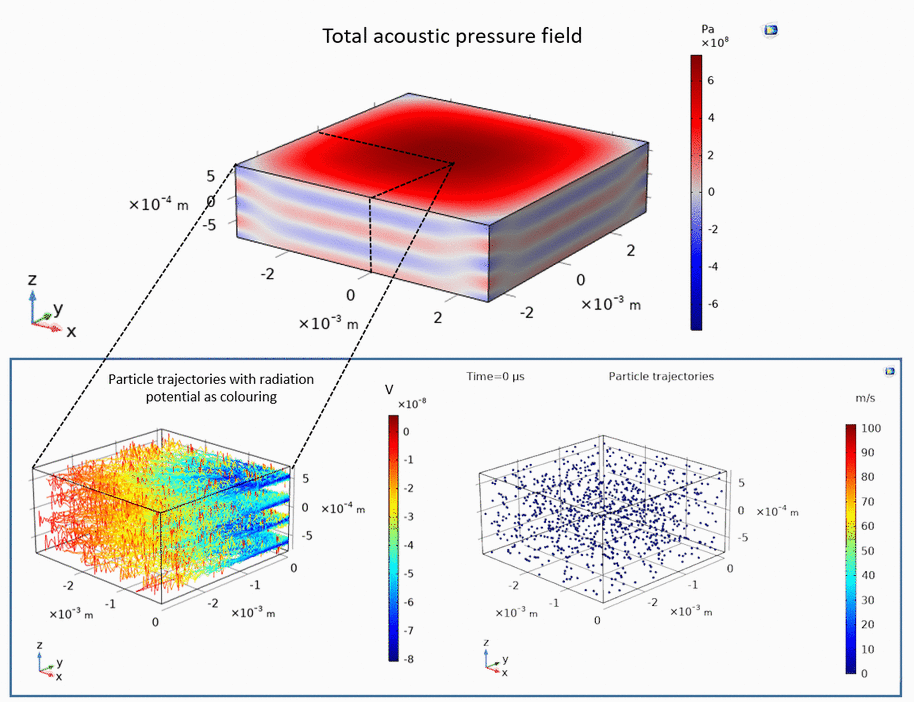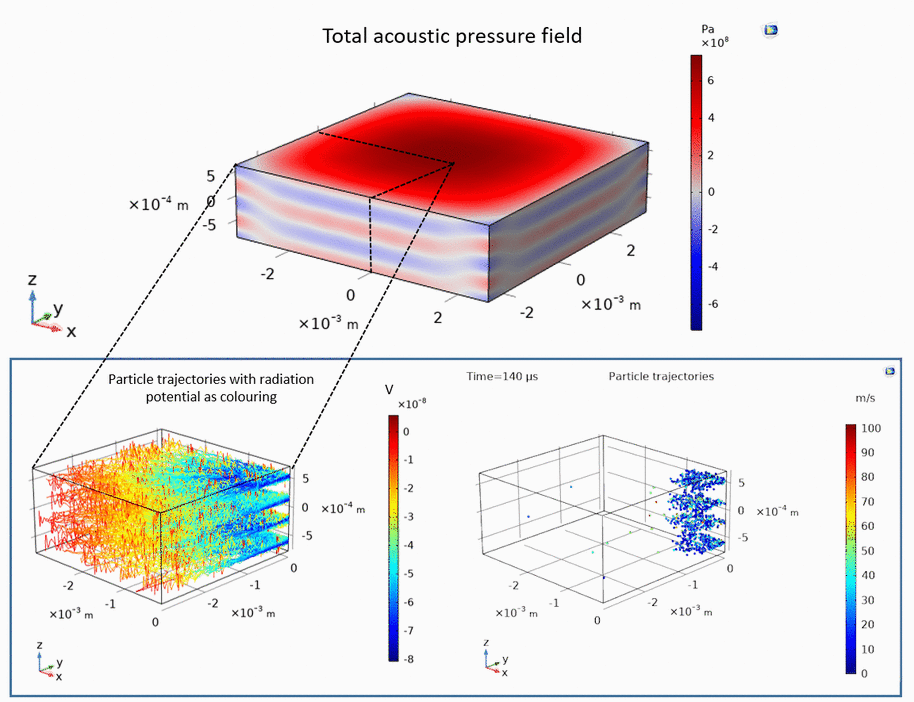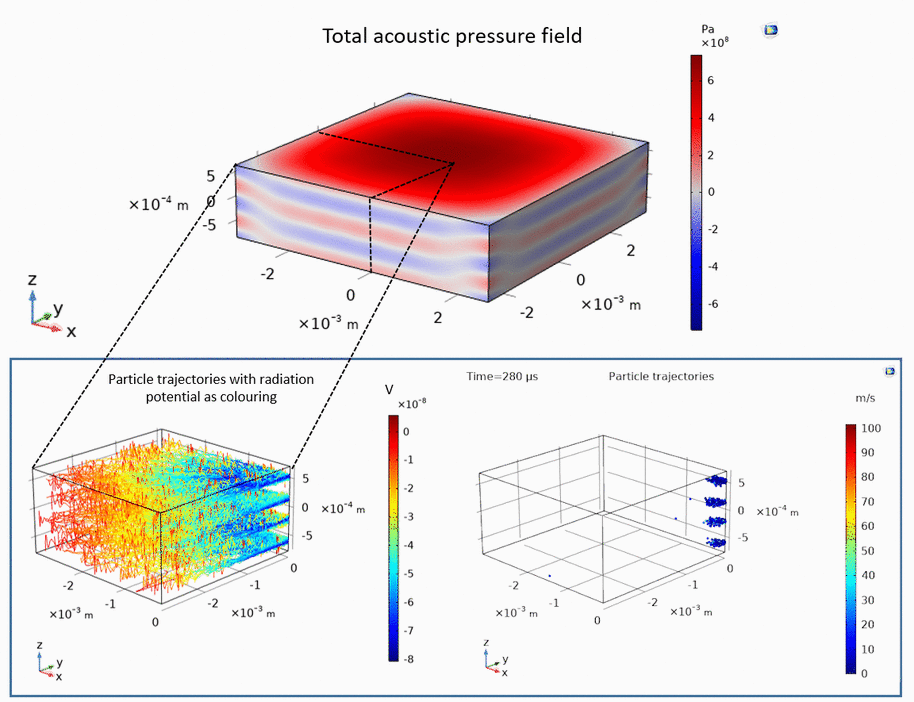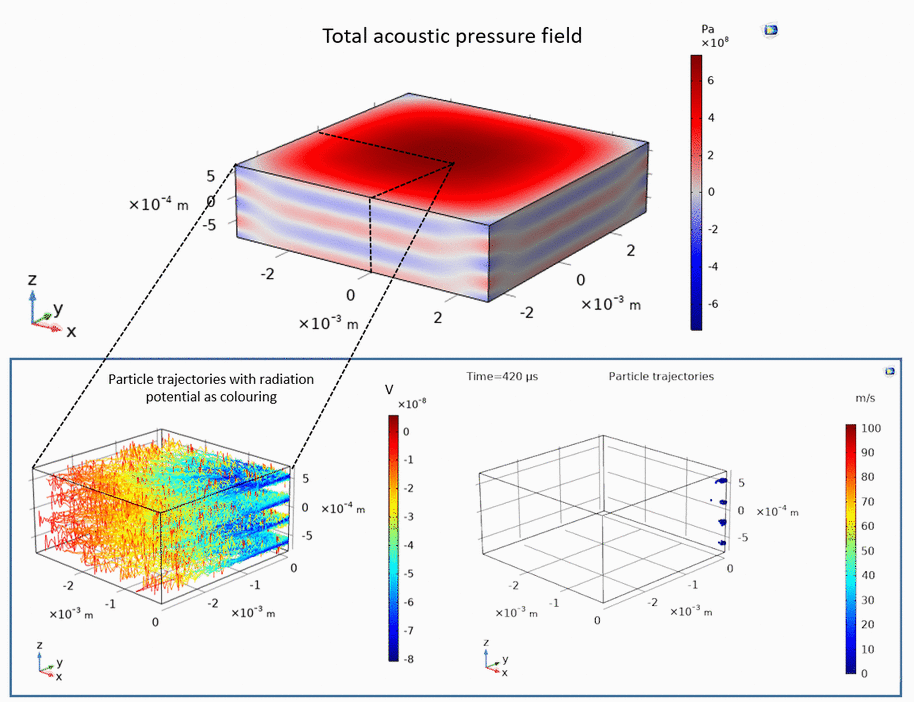
Publications
- Perra E, Lampsijärvi E, Barreto G, Arif M, Puranen T, Hæggström E, Pritzker KPH, Nieminen HJ. Ultrasonic actuation of a fine-needle improves biopsy yield. Sci Rep. 2021 Apr 15;11(1):8234. doi: 10.1038/s41598-021-87303-x.
- Pritzker KPH, Nieminen HJ. Needle biopsy adequacy in the era of precision medicine and value based health care. Archives of Pathology and Laboratory Medicine. 2019, In press.
- Veira D, Kekkonen T, Mäkinen J, Puranen P, Nieminen HJ, Kuronen A, Franssila S, Kotiaho T, Salmi A and Hæggström E. Practical realization of a sub-λ/2 acoustic jet. Scientific Reports, 2019, In press.
- Ylitalo T, Finnilä M, Gahunia HK, Karhula SS, Suhonen H, Valkealahti M, Lehenkari P, Hæggström E, Pritzker KPH, Saarakkala S, Nieminen HJ. Quantifying complex topology of degenerated articular cartilage surface by contrast-enhanced micro-computed tomography and parametric analyses. Journal of Orthopaedic Research, 2019, In press.
- Kauppinen S, Karhula SS, Thevenot J, Ylitalo T, Rieppo L, Kestilä I, Haapea M, Hadjab I, Finnilä MA, Quenneville E, Garon M, Gahunia HK, Pritzker KPH, Buschmann MD, Saarakkala S, Nieminen HJ. Morphometric analysis of calcified cartilage properties using micro-computed tomography. Osteoarthritis Cartilage. 2019, In press.
- Garcia-Perez A, Nieminxen HJ, Finnilä M, Salmi A, Pritzker KPH, Lampsijärvi E, Paulin T, Airaksinen AJ, Saarakkala S, Hæggström E. Delivery of agents into articular cartilage with electric spark -induced sound waves. Frontiers in Physics, 2018;6:116.
- Nieminen HJ, Laidmäe I, Salmi A, Rauhala T, Paulin T, Heinämäki J, Hæggström E. Ultrasound-enhanced electrospinning. Scientific Reports, 2018 13;8(1):4437. doi: 10.1038/s41598-018-22124-z.
- Kestilä I, Thevenot J, Finnilä MA, Karhula SS, Hadjab I, Kauppinen S, Garon M, Quenneville E, Haapea M, Rieppo L, Pritzker KP, Buschmann MD, Nieminen HJ, Saarakkala S. Osteoarthritis Cartilage. In vitro method for 3D morphometry of human articular cartilage chondrons based on micro-computed tomography. Osteoarthritis Cartilage. 2018 Aug;26(8):1118-1126. doi: 10.1016/j.joca.2018.05.012.
- Nieminen HJ, Barreto G, Finnilä MA, García-Pérez A, Salmi A, Ranjan S, Eklund KK, Pritzker KPH, Saarakkala S, Hæggström E. Laser-ultrasonic delivery of agents into articular cartilage. Scientific Reports. 2017 Jun 21;7(1):3991. doi: 10.1038/s41598-017-04293-5.
- Nieminen HJ, Gahunia HK, Pritzker KPH, Ylitalo T, Rieppo L, Karhula SS, Lehenkari P, Hæggström E, Saarakkala S. 3D histopathological grading of osteochondral tissue using contrast-enhanced micro-computed tomography. Osteoarthritis Cartilage. 2017 Oct;25(10):1680-1689. doi: 10.1016/j.joca.2017.05.021.
- Sakari S.Karhula, Mikko A.Finnilä, Jonathan D.Freedman, Sami Kauppinen, Maarit Valkealahti, Petri Lehenkari, Kenneth P.H. Pritzker, Heikki J. Nieminen, Brian D. Snyder, Mark W. Grinstaff, Simo Saarakkala.Micro-Scale Distribution of CA4+ in Ex vivo Human Articular Cartilage Detected with Contrast-Enhanced Micro-Computed Tomography Imaging.
- Karhula SS, Finnilä MA, Lammi MJ, Kauppinen S, Pritzker KPH, Nieminen HJ, Saarakkala. Effects of articular cartilage constituents on phosphotungstic acid enhanced micro-computed tomography imaging. PLOS One, 2017 30;12(1):e0171075. doi: 10.1371/journal.pone.0171075.
- Sundvik M*, Nieminen HJ*, Salmi A, Panula P, Hæggström E. Effects of acoustic levitation on the development of zebrafish, Danio rerio, embryos. Scientific Reports 2015;5:13596. * these authors contributed equally
- Nieminen HJ, Ylitalo Y, Karhula S, Suuronen J-P, Kauppinen S, Serimaa R, Hæggström E, Pritzker KPH, Valkealahti M, Lehenkari P, Finnilä M, Saarakkala S. Determining collagen distribution in articular cartilage using contrast-enhanced micro-computed tomography. Osteoarthritis and Cartilage. 2015;23(9):1613-21.
- Nieminen HJ, Ylitalo Y, Suuronen J-P, Rahunen K, Salmi A, Saarakkala S, Serimaa R, Hæggström E. Delivering agents surrogate locally into articular cartilage by intense MHz ultrasound. Ultrasound Med Biol. 2015 Aug;41(8):2259-65.
- Nieminen HJ, Salmi A, Karppinen P, Hæggström E and Hacking SA. The potential utility of high-intensity ultrasound to treat osteoarthritis, Osteoarthritis and Cartilage, 2014;22(11):1784-1799.
- Antila K, Nieminen HJ, Blanco-Sequeiros R, Enholm G. Automatic segmentation for detecting uterine fibroid regions treated with MR-guided High Intensity Focused Ultrasound (MR-HIFU). Medical Physics, 2014;41(7):073502.
- Kim YS, Trillaud H, Rhim H, Lim HK, Mali W, Voogt M, Barkhausen J, Eckey T, Köhler MO, Keserci B, Mougenot C, Sokka SD, Soini J, Nieminen HJ. MR thermometry analysis of sonication accuracy and safety margin of volumetric MR imaging-guided high-intensity focused ultrasound ablation of symptomatic uterine fibroids. Radiology, 2012;265(2):627-637.
- Venkatesan AM, Partanen A, Pulanic TK, Dreher MR, Fischer J, Zurawin RK, Muthupillai R, Sokka S, Nieminen HJ, Sinaii N, Merino M, Wood BJ, Stratton P: Magnetic resonance imaging-guided volumetric ablation of symptomatic leiomyomata: correlation of imaging with histology. Journal of Vascular and Interventional Radiology, 2012;23(6):786-794.
- Voogt MJ, Trillaud H, Kim YS, Mali W. P. Th. M., Barkhausen J, Bartels LW, Deckers R, Frulio N, Rhim H, Lim HK, Eckey T, Nieminen HJ, Mougenot C, Keserci B, Soini J, Vaara T, Köhler MO, Sokka S, van den Bosch MAAJ. Volumetric feedback ablation of uterine fibroids using magnetic resonance-guided high intensity focused ultrasound therapy. European Radiology, 2012;22(2):411-417.
- Lötjönen P, Julkunen P, Töyräs J, Lammi MJ, Jurvelin JS, Nieminen HJ: Strain-dependent modulation of ultrasound speed in articular cartilage under dynamic compression. Ultrasound in Medicine and Biology, 2009;35(7):1177-1184.
- Virén T, Saarakkala S, Kaleva E, Nieminen HJ, Jurvelin JS and Töyräs J. Minimally invasive ultrasound method for intra-articular diagnostics of cartilage degeneration. Ultrasound in Medicine and Biology, 2009;35(9):1546-1554.
- Kiviranta P, Lammentausta E, Töyräs J, Nieminen HJ, Julkunen P, Kiviranta I, Jurvelin JS. Differences in acoustic properties of intact and degenerated human patellar cartilage during compression. Ultrasound in Medicine and Biology, 2009;35(8):1367-1375.
- Kaleva E, Saarakkala S, Töyräs J, Nieminen HJ, Jurvelin JS: In vitro comparison of time-domain, frequency-domain and wavelet ultrasound parameters in diagnostics of cartilage degeneration, Ultrasound in Medicine and Biology, 2008; 34(1):155-159.
- Nieminen HJ, Julkunen P, Töyräs J, Jurvelin JS: Ultrasound speed in articular cartilage under mechanical compression, Ultrasound in Medicine and Biology, 2007;33(11):1755-1766.
- Kiviranta PJ, Töyräs J, Nieminen MT, Laasanen MS, Saarakkala S, Nieminen HJ, Nissi MJ, Jurvelin JS: Comparison of novel clinically applicable methodology for sensitive diagnostics of cartilage degeneration, European Cells and Materials, 2007;13:46-55.
- Nieminen HJ, Töyräs J, Laasanen MS and Jurvelin JS: Acoustic properties of articular cartilage under mechanical stress, Biorheology, 2006;43:523-535.
- Nieminen HJ, Saarakkala S, Laasanen MS, Hirvonen J, Jurvelin JS and Töyräs J: Ultrasound attenuation in normal and spontaneously degenerated articular cartilage. Ultrasound in Medicine and Biology, 2004;30:493-500.
- Nieminen HJ, Töyräs J, Rieppo J, Nieminen MT, Hirvonen J, Korhonen R and Jurvelin JS: Real-time ultrasound analysis of articular cartilage degradation in vitro. Ultrasound in Medicine and Biology, 2002;28:519-525.
- Töyräs J, Nieminen HJ, Laasanen MS, Nieminen MT, Korhonen RK, Rieppo J, Hirvonen J, Helminen HJ, Jurvelin JS. Ultrasonic characterization of articular cartilage. Biorheology 2002;39(1-2):161-169.
- Saarakkala S, Jurvelin JS, Zheng YP, Nieminen HJ, Töyräs J. Quantitative information from ultrasound evaluation of articular cartilage should be interpreted with care. Arthroscopy, 2007;23(10):1137-1138.
- Nieminen HJ, Zheng YP, Saarakkala S, Wang Q, Töyräs J, Huang YP, Jurvelin JS. Quantitative assessment of articular cartilage using high frequency ultrasound – research findings and diagnostic prospects, Critical Reviews in Biomedical Engineering, 2009;37(6):461-495.






